BT CORN: IS IT WORTH THE RISK?
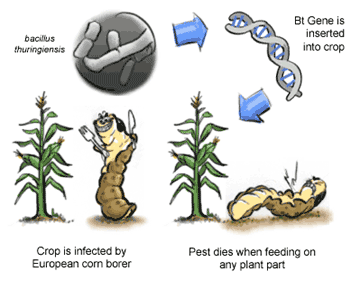
Bt corn, a genetically modified organism (GMO), has been both the poster-child and thorn-in-the-side of the plant biotechnology industry from the late 1990’s to present. There are several versions of this transgenic crop that each have a gene from an insect pathogen, Bacillus thuringiensis (Bt), which encodes a protein toxic to the European corn borer (ECB), an insect pest that eats and destroys corn stems (see Figure 1). Bt corn has proven effective in reducing crop damage due to ECB, yet public opposition to Bt corn has escalated amid fears of human health and environmental risks associated with the production and consumption of Bt corn.
History of Bt
Bt corn draws its humble origins from France, where in 1938 B. thuringiensis bacteria was grown in large quantities and sprayed on corn crops to prevent ECB damage[1]. Artificial selection of Bt strains has led to the successful targeting of many insect pests. Because no toxic effects of Bt on humans have been detected in its seventy years of use, it is now considered an acceptable pest control measure for the organic food industry[2]. To this day, Bt is an important part of many integrated pest management strategies. The success of the Bt spray has been limited because the bacteria cannot survive for very long on the plant’s surface. Bt is particularly ineffective at controlling ECB because these insect live most of their larval life inside the corn stem, not on the surface: sprays are only effective when the insects are starting its journey into the stem. Thus, a means of penetrating corn tissue with Bt is required to offer long-term anti-feeding measures against tunneling insects such as ECB.
Mechanism of Bt toxicity
Researchers investigated how this bacteria kills particular insects and discovered that Bt has two classes of toxins; cytolysins (Cyt) and crystal delta-endotoxins (Cry)[3]. While Cyt proteins are toxic towards the insect orders Coleoptera (beetles) and Diptera (flies), Cry proteins selectively target Lepidopterans (moths and butterflies). As a toxic mechanism, Cry proteins bind to specific receptors on the membranes of mid-gut (epithelial) cells resulting in rupture of those cells[4]. If a Cry protein cannot find a specific receptor on the epithelial cell to which it can bind, then the Cry protein is not toxic. Bt strains will have different complements of Cyt and Cry proteins, thus defining their host ranges[5]. The genes encoding many Cry proteins have been identified providing biotechnologists with the genetic building blocks to create GM crops that express a particular Cry protein in corn that is toxic to a particular pest such as ECB yet potential safe for human consumption.
Making Bt corn
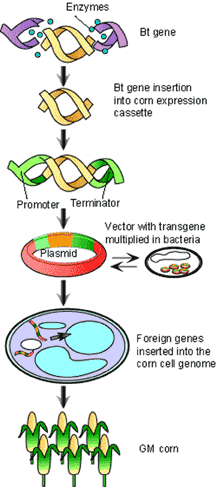
As it turns out, nature has its own biotechnologist called Agrobacterium tumefaciens which induces the growth of tumours on woody plants. These tumours are engineered by A.tumefaciens to produce a special food for the bacteria (opines) that plants normally cannot make. These tumours arise from a unique bacterial transformation mechanism involving the Ti-plasmid which coordinates the random insertion of a subset of its DNA (t-DNA) containing opine synthase genes into a plant chromosome[6] (see Figure 2). By replacing portions of the t-DNA sequence with genes of interest (such as Cry), researchers have been able to harness this transformational mechanism and confer new traits to many flowering plants including grasses such as corn7 and rice[8]. Cry-transformed corn varieties, called ‘Bt corn’, produce sufficient levels of Cry proteins to provide an effective measure of resistance against ECB and are now widely grown in North America.
Human health and environmental risks
The promise of this technology has been largely overshadowed by concerns about the unintended effects of Bt corn on human health and the environment. Cry protein toxicity, allergenicity, and lateral transfer of antibiotic-resistance marker genes to the microflora of our digestive system threaten to compromise human health. Despite these alarming possibilities, the risks to human health appear small based upon what is known about the bacterial endotoxin, its specificity, and confidence in the processes of plant transformation and screening[9]. The task of determining the levels of such risks, however, are immense. Human diets are complex and variable. How can we trace the acute or chronic effects of eating GM ingredients when they are mixed in with many other foods that may also present their own health hazards? It is even more complicated to determine the indirect risk of eating meat from animals raised on transgenic crops. These tests take time, and the results of clinical trials are not always clear-cut. It will likely take decades before we can know with any certainty if Bt corn is as safe for human consumption as its non-GM alternatives[10].
We currently know very little about the actual ecological risks posed by Bt corn. Bt corn may be toxic to non-target organisms, transgenic genes may escape to related corn species, and ECB and other pests may become resistant to Cry proteins[11]. The alleged effect of Bt corn pollen on Monarch butterfly larvae has rocketed to the front pages of major newspapers around the world (ex. CNN). Some research has shown that Monarch butterfly larvae fed their normal diet of milkweed leaves suffer a significant decline in fitness when those leaves are dusted with Bt corn pollen (Losey et al. 1999). The methodology of this experiment, however, has been harshly criticized by members of the scientific community.
Most recently, the threat of Cry gene escape into wild populations has been substantiated by the discovery that artificial DNA from transgenic corn has been detected in traditional corn varieties in remote areas of Mexico (Quist and Chapela, 2001). However, this study was pulled from NATURE magazine in an unprecedented fashion following a heated scientific and political debate[12]. While few contest that such transgenes are present in the local corn races of Mexico, there is still no evidence to suggest that these genetic constructs are “escaping” to become established in local corn races. We are limited to an educated guess as to the likelihood and speed of such genetic pollution[13].
Balancing risk and benefit
Despite the lack of conclusive evidence that GM foods present considerable risk to human health and environment, widespread use of this new technology is being compared to past mistakes such as broadcast spraying of populated towns with DDT to control mosquitoes during the 1950s. Notions of “frankenfoods”[14] and “agroterrorism”[15] corrupting our planet present theoretical possibilities that cannot be discounted given the remarkable ability of the unlikely to become an actuality. In truth, we must plead ignorance of the long-term impacts of GM crops[16].
Arguably, every food in our current diet carries with it associated risks, determined through “trial-and-error” extending back before to our hunter-gatherer origins. Often, we will accept a certain degree of exposure to known hazards to receive known benefits. Bt corn has obvious benefits for agricultural production, increasing profit margins through more efficient and consistent corn production and improving the working environment for farmers through reduced exposure to pesticides. In a surplus market, these benefits may be passed on to the consumer as a grocery bill reduction. On a global scale, decreased crop losses due to herbivory may translate into improved world food supply since corn remains a major staple in the global diet. Ecosystems are not likely to benefit from ECB-resistant Bt corn propagation since this technology replaces a largely mechanical (non-chemical) control for ECB.
These benefits, real or imagined, have been used as leverage by Bt corn proponents in the argument to accept what they argue are minimal levels of health and environmental risk. Yet many consumer, civil rights, and environmental advocacy groups characterize such arguments as industry propaganda, asserting that corporate benefits should not out-weigh the undetermined human health, socioeconomic and environmental risks.
The relative ease in engineering Bt biopesticides into crops such as corn, cotton and rice, combined with the cost effectiveness of Bt crops for growers under threat of ECB, makes banning this technology in North America seem unlikely. This reality highlights the necessity for the research community to improve methods for assessing risks posed by GM crops. While some industry proponents may resist, it is ultimately the public’s responsibility to ensure that this new technology is properly managed in the context of other pest management methods that have their own set of risks and benefits.
Notes
Glossary
Artificial Selection – the encouragement of certain traits in an animal through selective breeding by humans, both intentional or unintentional
Ti plasmid – “tumour-inducing” plasmid: originally found in the bacterium Agrobacterium tumefaciens, this plasmid integrates into a host cell genome and causes galls on plants. Biotechnologists can take advantage of this integration to insert genes of their choice into plant cells.
Lateral transfer – also called horizontal gene transfer, the movement of genetic material from one organism to another other than from parent to offspring, and often across species, genus, or even domain.
Antibiotic resistance marker genes – genes that allow biotechnologists to distinguish between plants that have been modified properly and those that have not depending on their suceptibility to antibiotics.
Screening – the process of selection of desirable plants from a large population of transformants (different insertional events) with variation in trait depending on location and number of t-DNA insertions.
Herbivory – the consumption of plants by animals, in this case to the detriment of the plant (predation).
References
1. Van Frankenhuyzen, K. in Bacillus thuringiensis, An environmental biopesticide: Theory and practice (John Wiley & Sons, 1993).
2. Whalon, M.E. & Wingerd, B.A. Bt: mode of action and use. Arch Insect Biochem Physiol 54, 200-211 (2003).
3. Crickmore, N. et al. Revision of the nomenclature for the Bacillus thuringiensis pesticidal crystal proteins. Microbiol Mol Biol Rev 62, 807-813 (1998).
4. Dorsch, J.A. et al. Cry1a Toxins of Bacillus Thuringiensis Bind Specifically to a Region Adjacent to the Membrane-Proximal Extracellular Domain of Bt-R-1 in Manduca Sexta: Involvement of a Cadherin in the Entomopathogenicity of Bacillus Thuringiensis. Insect Biochemistry and Molecular Biology 32, 1025-1036 (2002).
5. De Maagd, R.A., Bravo, A. & Crickmore, N. How Bacillus Thuringiensis Has Evolved Specific Toxins to Colonize the Insect World. Trends in Genetics 17, 193-199 (2001).
6. Bevan, M.W. & Chilton, M.D. T-DNA of the Agrobacterium Ti and Ri plasmids. Annu Rev Genet 16, 357-384 (1982).
7. Ishida, Y. et al. High efficiency transformation of maize (Zea mays L.) mediated by Agrobacterium tumefaciens. Nat Biotechnol 14, 745-750 (1996).
8. High, S.M., Cohen, M.B., Shu, Q.Y. & Altosaar, I. Achieving successful deployment of Bt rice. Trends Plant Sci 9, 286-292 (2004).
9. Kuiper, H.A., Kleter, G.A., Noteborn, H.P. & Kok, E.J. Assessment of the food safety issues related to genetically modified foods. Plant J 27, 503-528 (2001).
10. Sudakin, D.L. Biopesticides. Toxicol Rev 22, 83-90 (2003).
11. Sharma, H.C. & Ortiz, R. Transgenics, Pest Management, and the Environment. Current Science 79, 421-437 (2000).
12. Ochert, A. Caught in the maize at Berkeley. California Monthly (2002).
13. Letourneau, D.K., Robinson, G.S. & Hagen, J.A. Bt crops: predicting effects of escaped transgenes on the fitness of wild plants and their herbivores. Environ Biosafety Res 2, 219-246 (2003).
14. Golden, F. Who’s afraid of Frankenfood? Time 154, 49-50 (1999).
15. van Bredow, J. et al. Agroterrorism. Agricultural infrastructure vulnerability. Ann N Y Acad Sci 894, 168-180 (1999).
16. Hoffmann-Riem, H. & Wynne, B. In risk assessment, one has to admit ignorance. Nature 416, 123 (2002).
(Art by Jiang Long and Jen Philpot)