EVOLUTION OF ALCOHOL SYNTHESIS
We need to appreciate beer more. Alcohol has a long history in human affairs, and has been important in purifying and preserving food and drink, and in making our parties livelier. We owe it all to a tiny little microorganism, Saccharomyces cerevisiae, which converts complex plant sugars into smaller, simpler, more socially potent molecules of ethanol. This is a remarkable process that seems to be entirely to our benefit (it has even been argued that beer is proof of the existence of God*), but recent research has shown that the little buggers do it all entirely for their own selfish reasons, and they’ve been busily making alcohol that has gone undrunk by humankind for tens of millions of years.
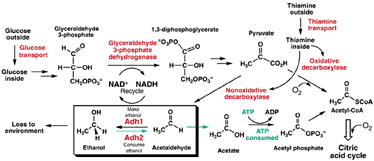
In order to explain how we know this, forgive me, but I must explain some very basic biochemistry, and summarize what cells do to extract energy from sugar. We start with a 6 carbon sugar molecule. As a first step, called glycolysis, enzymes in the cell snap the molecule in half, liberating a little bit of energy and producing two 3-carbon molecules, called pyruvate.
Pyruvate gets passed on to the next step, called the citric acid cycle. This is a series of reactions that breaks the 3-carbon chain down carbon by carbon, liberating yet more energy at each step. It’s all the steps after glycolysis that extract the bulk of the energy from the sugar molecule, but there’s a catch: these steps require oxygen to run (this is also called the aerobic pathway). No oxygen, no citric acid cycle. Glycolysis can run, but some of the reaction products (especially a compound called NADH) accumulate, and soon enough that reaction would get choked off, too.
In the absence of oxygen, cells can continue to get that little bit of energy from glycolysis if only they can get rid of the accumulating reaction products somehow. In us, our cells do that by carrying out an additional reaction to convert excess pyruvate and NADH to another 3-carbon molecule, lactate, and NAD+. Lactate diffuses out of the cells and into the blood stream, forming lactic acid. When you are exercising anaerobically, that is, making your cells work harder than you can deliver oxygen to them, they limp along by dumping 3-carbon molecules in the form of lactic acid so they can keep burning sugar inefficiently. Once you’re done working out, and your oxygen intake catches up, the lactate is converted back to pyruvate and can be burned completely and efficiently in the citric acid cycle.
Yeast do something different. If they are under anaerobic conditions, say, deep in the flesh of some decaying fruit, or in a wine bottle, they have the same problem: they want to keep their metabolism going by carrying out glycolysis, but to do that they have to get rid of accumulating products, somehow. They don’t do it by making lactic acid, though (thank goodness—if they did, fermentation would produce a vinegary acid). Instead, they take the 3-carbon pyruvate and split off one carbon, producing CO2, which is given off as a gas. Any homebrew beer makers out there will be familiar with the idea of monitoring fermentation by observing the gas being produced.
The 2-carbon molecule left behind is called acetaldehyde. Acetaldehyde is further processed by an enzyme called alcohol dehydrogenase, Adh for short, which also recycles NADH. Adh converts the 2-carbon acetaldehyde into another 2-carbon molecule, ethanol. Alcohol. Booze.
Just like us, yeast produce this byproduct to keep going under anaerobic conditions, and when oxygen is available, they try to recover the energy in the alcohol. Familiar brewers’ yeast has two forms of alcohol dehydrogenase: Adh1, which favors the production of alcohol from acetaldehyde, and Adh2, which more effectively runs the reaction in reverse, producing acetaldehyde from alcohol, and allowing the 2-carbon molecule to be fed back into the citric acid cycle.
If you’d rather see this in a simple biochemical diagram of the yeast pathways below: it says the same thing I just wrote up there.
The second line of evidence is very cool. It would be instructive to be able to directly examine the metabolism of yeast from 80 million years ago, and measure for ourselves the activity of their Adh enzyme. We don’t have a time machine, unfortunately, but we do have the ability to reconstruct ancient genes.
The authors compared the sequences of Adh1 and Adh2 from S. cerevesiae and from 15 other Adh homologs in other yeast species. They then calculated the maximum likelihood gene sequence for the last common ancestor of these enzymes, the primordial alcohol enzyme, which they called AdhA. They then took modern yeast, removed their Adh1 and Adh2 genes, and replaced them with AdhA. Voilà, they have yeast from the Age of the Dinosaurs.
They then analyzed the chemical kinetics of this enzyme. The question was whether it was more like Adh1, the enzyme that primarily makes ethanol, or whether it was more like Adh2, the enzyme that primarily consumes alcohol. Did yeast evolve this enzyme to make a byproduct to inhibit its competitors, or did it evolve it to eat this byproduct?
The answer is that it was more like Adh1, and that early yeast were brewers, not drinkers.
“Notably, the kinetic properties of the remaining ancestral AdhA candidates resembled those of Adh1 more than those of Adh2. From this, we inferred that the ancestral yeast did not have an Adh specialized for the consumption of ethanol, similar to modern Adh2, but rather had an Adh specialized for making ethanol, similar to modern Adh1. This suggests that before the Adh1-Adh2 duplication, the ancestral yeast did not consume ethanol. This implies that the ancestral yeast also did not accumulate ethanol under aerobic conditions for future consumption and that the make-accumulate-consume strategy emerged after Adh1 and Adh2 diverged. These interpretations are robust with respect to the ambiguities in the reconstructions.”
We can assemble a history of yeast fermentation from this information now. The first step was the gradual evolution of efficient alcohol-producing enzymes that allowed the yeast to colonize and exploit rotting fruit exclusively. This occurred a very long time ago, in the Cretaceous. Next, there was a gene duplication event that produced two copies of Adh; initially, both would have done exactly the same thing, just allowing the lucky duplicators to pump out alcohol even faster. With two copies, though, one would have more freedom to drift and change its enzymatic properties without serious consequence to the owner. One fortuitous change would be a shift in enzyme kinetics in one copy to better promote conversion of alcohol back to acetaldehyde and enter back into the citric acid cycle. So, first they learned how to make an environmental poison to give them exclusive access to a food source, and then that same machinery was adapted to better allow them to eat that poison, permitting them recover some of the energy lost in secreting it.
Notes
* “Beer is proof that God loves us and wants us to be happy,” Benjamin Franklin.
References
Thomson JM, Gaucher EA, Burgan MF, DeKee DW, Li T, Aris JP, Benner SA (2005) Resurrecting ancestral alcohol dehydrogenases from yeast. Nature Genetics 37:630-635.
Woolfit M, Wolfe K (2005) The gene duplication that greased society’s wheels. Nature Genetics 37:566-567.