HUMAN CLONING: SCIENCE FICTION OR REALITY?
(August 2004)
On 27th December, 2002, Dr. Brigitte Boissilier, scientific director of Clonaid, claimed the birth of the first cloned human. The announcement was greeted with revulsion from the general public and skepticism among the scientific community. Although the world still awaits scientific verification of this procedure, the claim intensified the ongoing public debate regarding this technology and its potential uses. Cloning has been hypothesized to have the potential to revolutionize at least two aspects of human life.
Therapeutic cloning aims to produce embryonic stem cells which, because of their multipotentiality, have been proposed as a source of cells for the replacement of damaged or defective tissue in adults. In this form of cloning, cells are never allowed to develop into a complete organism. Reproductive cloning, on the other hand, aims to produce offspring with similar genetic identity to previously existing organisms. This practice raises many ethical and moral questions.
Embryonic Stem Cells: A Cure for many diseases?
Several adult tissues, once damaged, are not able to repair themselves. The neural system is the most obvious example of this. When someone sustains spinal chord injury, the body is unable to repair the neurons in the spinal chord and the person remains paralyzed for life. This is in direct contrast to other tissues such as the blood and skin, where millions of cells are lost and replaced each day. Stem cells, which have the ability to become these mature or differentiated cells, are responsible for maintaining tissues at constant levels. Unfortunately, stem cells are generally difficult to find in adults. When they are isolated, they are often hard to grow into cell lines. It is unlikely that adults will be a sufficient source of stem cells for therapeutic purposes. Embryonic stem cell lines have the potential to divide indefinitely and are pluripotent (they have the potential to differentiate into various types of cell). An important consideration in therapeutic cloning is the possibility that the patient’s immune system will reject the transplanted cells. Each patient is an ideal donor for himself or herself because this abolishes the need to find a donor with compatible tissues. Cloning the individual to create unlimited stem cells would be an ideal way to circumvent this situation.
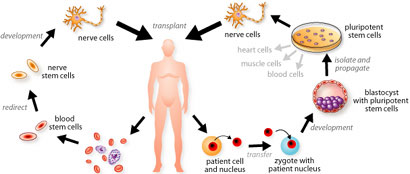
If research with embryonic stem cells continues, the source of these cells will become an issue. Embryonic stem cells are taken from the early embryo, shortly after fertilization. At present, embryos that are frozen for couples undergoing in vitro fertilization are used to collect embryonic stem cells. Generally, due to the high failure rate of the procedure, multiple eggs are fertilized and stored. These are then transplanted into the mother one at a time until she becomes pregnant. The remaining embryos remain in storage and are generally forgotten. They present a potential limited source of stem cells as long as the issue of tissue rejection is not addressed.
More recently a new technique has been developed called somatic cell nuclear transfer (SCNT). SCNT involves the removal of an egg from a hormonally treated female. The donor egg is enucleated and the nucleus of a somatic cell is transferred into its cytoplasm (nuclear transfer). The egg is then stimulated to start dividing. After sequential cell divisions, the blastocyst formed contains cells that will give rise to all tissues in the embryo. The blastocyst can be used to generate embryonic stem cells, which are genetically identical to the somatic cell donor, thereby eliminating the possibility of rejection when transplanted.
Reproductive cloning
Alternatively, the blastocyst can be implanted back into a female and brought to term resulting in a cloned organism. This procedure was allegedly used in the production of the first cloned human mentioned above.
SCNT was pioneered by Dr. Ian Wilmut at the Roslin Institute in Scotland. He used this technology to produce the world’s first cloned sheep, Dolly, in 1996. Unfortunately, she developed arthritis early in life and showed premature signs of aging. Dolly was euthanized in February 2003 after doctors detected a progressive lung disease. She had only reached half the lifespan of a typical sheep and, while it has not been proven that her cloned status played a part in her early death, scientists remain sensitive to the fact that the full implications of this technology are not yet known. Since 1997, this technology has been used to clone pigs, mice, goats and cats. In November 2001, the production of the first human embryo by this method was reported in the Journal of Reproductive Medicine.
Human reproductive cloning has been considered unethical by the scientific community due to safety considerations. According to animal studies, production of a healthy reproductive clone will create potential morbidity and mortality in women carrying cloned fetuses, newborns and egg donors. In animal studies, only 1-2% of cloned zygotes survive to the birth stage. Therefore, a large number of eggs are needed to get a successful birth. In order to obtain enough eggs for one successful birth, many women will need to donate eggs. Studies on in vitro fertilization show that there is a risk of ovarian hyperstimulation associated with egg donation, which can sometimes cause maternal death.
There are also complications involved in carrying a cloned animal to term. High rates of abortion and neonatal loss have been observed. There have also been many late gestational abortions. This is in contrast to the early spontaneous abortion that occurs naturally in humans during the first trimester. Fetal abnormalities are also abundant; abnormal placenta, pregnancy toxemia, hydroallantois (excessive fluid accumulation) all frequently occur and result in fetal abnormality. Consequently, newborns often die. High rates of late gestational losses, including stillborns and children with severe health problems would have a negative psychological effect on mothers. Furthermore, in the event that an infant is born, there is a high probability that it will not be healthy. A wide variety of tissues and organs fail to develop properly. Many cloned animals have been born with abnormalities such as respiratory failure, immune deficiency, inadequate renal function, cardiovascular problems, large birth size, post natal weight gain, and liver and joint defects. In addition to these problems, human clones would have a risk of aberrant brain development. Brain development problems in animals could easily have gone undetected in the past, and human brain development is even more complex than that of animals. This list of defects cannot be diagnosed or prevented using current technology of prenatal screening or ultrasonography. The possible benefits of human reproductive cloning do not seem to justify the risks to all the parties involved.
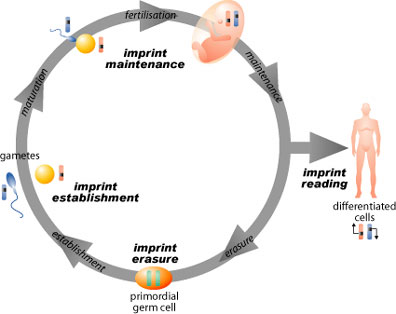
The current problems with mammalian reproductive cloning are likely related to faulty epigenetic reprogramming. Reprogramming is the process by which DNA and its associated proteins (in a nucleus transplanted from a somatic cell) are reset to coordinate early developmental processes. When SCNT is performed, the adult nucleus needs to respond to the egg cytoplasm as though it were a nucleus of a zygote. Theoretically, this will shut off the genes that are active in adult cells, which would normally be inactive in a zygote. Reprogramming needs to occur in a relatively short time, a few days in most mammals. In sexual reproduction, reprogramming is not necessary as genes are derived from germ cells. The egg and sperm are preprogrammed during the egg and sperm development and continues throughout early development. Programming is thought to occur by imprinting.
![methylation[1]-GIF.gif](https://www.scq.ubc.ca/wp-content/uploads/2006/08/methylation[1]-GIF.gif)
Imprinted genes have a “mark” imposed on or near them. The most well studied mode of “marking” is methylation. Imprinting patterns are distinct in maternal and paternal genes. This causes differential behavior of genes depending on whether it has maternal or paternal imprinting. Thus, the behavior of the allele is different depending on whether the chromosome came from the father or the mother. Mouse studies in which an embryo was created from two maternal derived gametes resulted in fetal abnormality and death. In humans, mutations that disturb the inactivation of genes can result in tumors in both children and adults. Some well known genetic disorders in children are caused by mutations in regions of DNA methylation, the imprint control regions. These diseases, including Angelman Syndrome and Beckwith-Weidemann Syndrome, result in a combination of mental retardation and congenital abnormalities.
Nature versus nurture
There is a common perception that cloning an individual will create someone identical in every physical way. However, the clone will be distinct from the nuclear donor in many respects. Observations from human twins (natural clones) and cat clones have shown that identical genetic material is not the only determinant of character and appearance. Dissimilarities in human clones will be caused by different mutations, stochastic development, various imprinting effects, differing environmental and nutritional inputs while in the uterus and different societal inputs. Cloning will not necessarily reproduce the character, appearance and physiology of the nuclear donor. Studies with the cat Rainbow and her cloned copy (named Cc for carbon copy) are a good demonstration of this.
The cloning of Rainbow has demonstrated that the phenotype of the individual is largely determined by environmental factors. This project was funded by Genetic Savings & Clone, a company interested in cloning pets, and performed by researchers at Texas A&M University. Their paper, published in Nature in 2002, presented the details of their project, including DNA evidence that showed Cc was a clone. Interestingly, Rainbow and Cc have difference coat color and patterning, personalities and body type. Rainbow is reserved while Cc is curious and playful; Rainbow is chunky while Cc is sleek.
There are practical purposes for animal reproductive cloning. Livestock, such as cattle, are a prime target for reproductive cloning. It allows breeders to avoid the randomness of natural sexual reproduction. Consistent replication of existing favorable trait combinations such as efficient growth and high milk production is possible.
Current attempts to clone a human
Allegedly, there has been some progress in producing human embryos for therapeutic purposes. There has not, however, been any good scientific evidence presented to confirm these experiments. Nuclear transfer was reported with human skin cells in the Journal of Reproductive Medicine in 2001. The embryo needs to reach the 64 cell stage before it can be used to derive embryonic stem cells. It is still considered scientifically significant that one cell multiplied to the 6 cell stage. This could represent progress in human reproductive cloning. The validity of the experiments has been questioned due to three scientists resigning from the editorial board of the journal because they believed there was insufficient experimental validation. The authors failed to provide evidence that DNA from dividing cells came from the donor cell or that the cells were functional.
Several people claim that they have gone farther than the early embryo stage and have successfully cloned a human, others are currently attempting to do so. As mentioned earlier, Dr. Brigitte Bosselier from Clonaid, claims to have cloned three human babies. Clonaid is a company funded by the Raelian Movement that is devoted to creating human clones. They have yet to provide DNA samples or any other reliable evidence to support these claims. Clonaid says it has a list of 2,000 people willing to pay $200,000 to have themselves or a loved one cloned. Dr. Severino Antinori and his collaborator, Dr. Panos (Panayiotis) Zavos announced in Nov. 2001 that 200 couples in an un-named European country would attempt to give birth to the first human clone. The births were expected to occur in the second-half of 2002, presumably following the traditional nine-month gestation period. No results have been announced.
Legislation
Despite a lack of evidence to show that a human has been cloned, announcements by the Raelians and the cloning of Dolly the sheep have resulted in the development of several laws. In the United States, Congress has passed a bill that would ban all human cloning (this bill has yet to be passed by the senate). Most countries, including Canada, do not have specific cloning laws. Canada is in the midst of debating Bill C-13, the Assisted Human Reproduction Act. The Council of Europe has signed a protocol banning human reproductive cloning research defined as “any intervention seeking to create a human being genetically identical to another human being, whether living or dead.” The interpretation of “human being” according to domestic laws will be used to decide if this includes stem cells. Moreover, UK, Ireland and Germany did not sign this protocol but they previously had laws or they have implemented their own laws. Ireland and Germany have forbidden research on embryos for many years. The UK has banned reproductive cloning but SCNT for experimental treatment is not prohibited. Human cloning claims have spurred governments to create regulations governing human cloning before rather than after the laws are required.
Conclusion
As of yet, human reproductive cloning still seems to be science fiction. Current medical and scientific evidence suggest that it is dangerous to both mother and child. Successful reproduction of a healthy human child is unlikely and not worth the dangers involved. The current claims made by religious factions are likely false. They have, however, served the purpose of motivating governing bodies and society to contemplate issues related to both therapeutic and reproductive cloning.
References
1. Wilmut I, Schnieke AE, McWhir J, Kind AJ, Campbell KHS. Viable offspring derived from fetal and adult mammalian cells. Nature 1997; 385: 810-813.
2. Rideout WM, Eggan K, Jaenisch R. Nuclear Cloning and Epigenetic Reprogramming of the Genome. Science 2001;  293: 1093-1098.
3. Shin T, et al. A cat cloned by nuclear transplantation. Nature 2002; 415: 859.
4. Humphreys D, et al. Abnormal gene expression in cloned mice derived from embryonic stem cell and cumulus cell nuclei. PNAS 2002; 99:12889.
5. Schatten G, Prather R, Wilmut I. Cloning Claim Is Science Fiction, Not Science. Science 2003; 299: 344.
Additional Reading
1. The National Academies. (2002). Scientific and Medical Aspects of Human Reproductive Cloning. Washington DC, National Academy Press.
2. Lauritzen, P., Ed. (2001). Cloning and the Future of human Embryo Research. New York, New York, Oxford University Press.
4. http://www.who2.com/brigitteboisselier.html
(Art by Jen Philpot)