THE UNACKNOWLEDGED WORKERS OF THE BIOTECHNOLOGY INDUSTRY: MICROBES
(August 2003)
Although seen by many as a development of the 20th century, biotechnology was, in fact, one the first technologies developed, and the earliest examples of these technologies are still the most economically important. Early use primarily consisted of methods of fermentation, both for preserving foods, and for the production of alcoholic beverages. In spite of the length of time that has passed since biotechnology came into use, it is only in the past century that we have begun to understand how these innovations work and how they can be improved. Furthermore, great strides have been made in developing new, genetically-modified microbes for these applications.
Fermentation is perhaps best described as the conversion of sugars to ethanol. It occurs via the following pathway:
During the ferment, many other byproducts of metabolism are produced. It is the sum of all of these products that gives rise to the taste and appearance of the final product.
The Essential Ingredient in Beer
The first archealogical evidence of beer production comes from ~6000 BC in ancient Babylon [1]. The discovery of how to brew beer was likely an accident, resulting from harvested barley becoming wet and being left for a time. This mix would have fermented and become a crude form of beer. It is likely that the origins of wine are similar, but with grape juice substituted for the wet barley.
Modern beer production consists of several steps, first allowing the barley to germinate [2]. This allows the starch in the barley to be degraded to sugars by enzymes in the grain. Following germination, the barley is heated to kill the sprouted plants. At this point, water and flavourings such as hops are added and the mix, or mash, is boiled to extract sugars and flavours from these ingredients. The solid particles are then filtered out of the liquid extract, or wort. Yeast is then added to the mix and is left to ferment. Although in modern beer making the strain of yeast used is pre-selected and artificially introduced, in medieval times, the yeast was carried into the mix by winds, and each beer could have several strains of yeast in it. There are still some regions where beer is produced in this manner, and this style of brewing is referred to as lambic brewing [3].
The first modern studies on beer production were conducted by the father of microbiology, Louis Pasteur. Pasteur studied fermentation, and discovered that all fermented materials contained yeast. He also found that spoiled batches of beer occurred as a result of bacterial contamination of the ferment, and this contamination could be prevented by heating the wort to 55°C before adding the yeast. This is the process of pasteurization, named after Pasteur, which is still used today. His 1876 work “Etudes sur la Biere” summarized the results of much of his work.
Today, there are two types of yeast used in beer production. The first is known as top-fermenting yeast and are used in the production of ales. Ales include a number of different styles, such as pale ales, porters, stouts, and witbiers. The other type of yeast is the bottom-fermenting yeast, used for lager production. Top fermenting yeasts are members of the species Saccharomyces cerevisiae, which are also used for the production of bread. This yeast typically ferments at relatively high temperatures (16°C-25°C). Lagers are produced by the action of Saccharomyces carlsbergensis and fermentation occurs at lower temperatures (4°C-9°C). Examples of lagers include pilsener, bock, and dunkels. There are thousands of different strains of these types of yeast used in beer production. One yeast strain used for the production of beer and wine, Saccharomyces cerevisiae, has had its genome completely sequenced. It is hoped that by using the information from the genome sequence, that better tasting beers and wines can be developed.
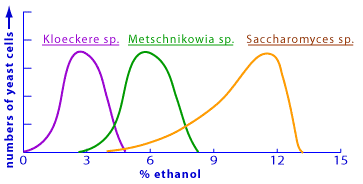
There are 15 different yeast genera that are generally accepted as a part of wine-making (oenology), and include the following species: Brettanomyces and its sexual equivalent Dekkera; Candida; Cryptococcus; Debaryomyces; Hanseniaspora and its asexual counterpart Kloeckera; Kluyveromyces; Metschnikowia; Pichia; Rhodotorula; Saccharomyces; Saccharomycodes; Schizosaccharomyces; and Zygosaccharomyces [4]. Of the yeast found on the grapes themselves, Kloeckera and Hanseniaspora make up 50-75% of the total yeast population. Industrial winemaking requires careful selection of the yeast used in fermentation, as each yeast strain imparts certain flavour characteristics on the final product.
Depending on whether the wine being produced is white or red, the skins will be left in the juice during the fermentation or will be removed. The red colour of red wine comes about primarily from the skin. If cultured yeast (guided fermentation) are going to be used during the fermentation, they will be added at this point. The cultures used for inoculation are primarily Saccharomyces sp., similar to those used in beer production.
On Using Saccharomyces…
The reasons for the use of Saccharomyces in beer and wine production are relatively straightforward. Both beer mash and grape juice are mixtures characterized by a high concentration of sugar and low pH. During fermentation these sugars lead to the production of ethanol and carbon dioxide. It is important that the yeast strain used is able to survive the higher ethanol concentrations produced. For beer, these concentrations generally range from 3-9%, while for wine they are substantially higher at 11-15%. Saccharomyces strains are fairly resistant to high ethanol concentrations. Even when present as a very small percentage of the initial yeast population, these strains tend to be the dominant strain at the end of natural fermentation due to their ethanol tolerance [4].
Saccharomyces strains are also chosen based on their ability tolerate other compounds, such as sulphite [4]. In modern winemaking, the native yeast strains are not killed by heat as they are in beer-making, because the act of heating alters the final taste of the wine. Instead, the grape juice is treated with sulphite to remove the native strains, thus making it necessary for the Saccharomyces strains to survive the addition of these compounds.
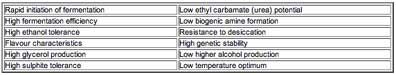
In addition to these practical reasons for choosing Saccharomyces strains, vintners may also want to impart a particular characteristic to the final wine produced. For this, the winemaker may select strains that have been characterized in the past as having a particular taste. Thus, there is a great deal of trial and error in selecting yeast strains. It is also not yet entirely clear what genes are responsible for giving a particular yeast strain a particular flavour in the final product. A partial list of desired characteristics in wine is given in the table below.
Wine Production Can Mean Twice the Fermentation
Following the primary fermentation of wines, several secondary fermentations may be carried out, depending on the final product desired. During the production of many wines, two main acidic components are present: malic and tartaric acid. The final concentration of these organic acids determines the final acidity of the wine produced [4]. Quality wines may undergo a secondary fermentation, known as malolactic conversion, to decrease the acidity of a wine. In this process, a lactic acid bacterium known as Oenococcus oeni is introduced into the grape juice and catalyzes the conversion of malic acid to the less acidic-tasting lactic acid [4]. This conversion is important not only because it improves the palatability of a wine, but it can increase shelf-life as well. One problem with this process is that this secondary fermentation often fails to progress very quickly. This leads to increased spoilage products in the wine, and and in particular produces biogenic amines, the main culprit behind wine-induced headache [5].
Several non-desirable metabolites can be produced during wine fermentation, such as biogenic amines, as mentioned above. Biogenic amines, which are byproducts from the metabolism of amino acids [5], are vasodilators, and therefore can induce headaches in approximately one in three wine-drinkers. Urea is also produced during the fermentation. Excess urea in wine can lead to the production of ethyl carbamate, a known carcinogen. Research being conducted at UBC is currently working on these problems by developing yeast strains that can carry out the malo-lactic conversion, without the need for a second, inconsistent fermentation and strains that are capable of degrading urea to harmless compounds [6, 7].
Different types of secondary fermentations are also carried out in the production of sparkling white wines and sherry. For champagne, this consists primarily of adding fermentable sugar to the bottle when the wine is added. Following corking, fermentation takes place in the bottle, producing more ethanol and carbon dioxide. The carbon dioxide dissolves into the wine, producing the carbonation observed when uncorked. Sherry involves an interesting process, whereby a stack of barrels, known as a solera, are used. The wine base (product of the primary fermentation) is added to the top barrel and allowed to age for a period of several months. Then half of the wine from the top barrel is added to the wine in the barrel below it. This is continued every few months until the wine in the bottom barrel is ready for bottling. This process permits greater oxygenation of the wine. Higher oxygenation is required with sherry wines to promote the growth of a flor, or film, of yeast on the barrels and on the surface of the wine [8]. The yeast strains involved forming the flor are Torulaspora fermentati, Zygosaccharomyces rouxii, and Saccharomyces cerevisiae. This combination of yeasts produces acetaldehyde and glycerol at higher concentrations than in the primary ferment, resulting in the distinct flavour of sherry. At the end of the entire process, the wine may have spent up to 10 years in a solera.
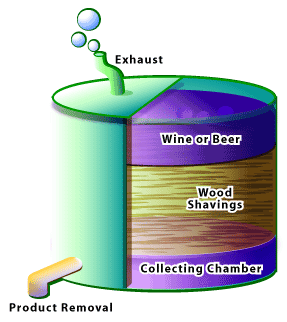
Wine and beer are both used in the production of another important food product: vinegar. This process requires that a vinegar generator first be created (Figure 2). This consists of a large wooden vat, approximately 8 metres in height and 4 metres in diameter, which is loosely packed with wood shavings. The shavings act as a surface on which the bacteria can grow. An alcohol source is then poured over the wood shavings; beer is used if malt vinegar is desired, while wine is used to produce wine vinegar. Following its first run through the reactor, the liquid is collected and run through the generator several more times. When the acetic acid content rises above 4%, it can be collected and bottled as vinegar [2]. The oxidation of ethanol to acetic acid is carried out by Acetobacter sp. For the production of purified acetic acid or white vinegar, the acetic acid solution produced by this reactor is collected and distilled. The pure acetic acid is then diluted to the desired concentration to be sold as white vinegar. High-quality vinegars may be allowed to age, during which time esters form from the reaction of acetic acid and residual ethanol. This allows the formation of very complex mixtures of flavours that are not found in the beer or wine alone.
Despite the fact that these technologies have been with humanity for millennia, they are still among the most economically important products of biotechnology. Annual beer production is now over 1 billion barrels, while for wine it is over 600 million barrels. These represent billions of dollars of sales and tens of thousands of jobs. Research is seeking to make the products of beer and wine production even better and safer than they have ever been before.
References
1. Brown, C.M., Campbell, I. and F.G. Priest. 1987. Introduction to Biotechnology. Blackwell Scientific Publications, Oxford.
2. Brock, T.D., Madigan, M.T., Martinko, J., and J. Parker. 1994. Biology of Microorganisms. Prentice Hall, New Jersey.
3. http://www.beer-brewing.com
4. Pretorius, I. 2000. Tailoring wine yeast for the new millennium: novel approaches to the ancient art of winemaking. Yeast. 16:675-729.
5. Lonvaud-Funel, A. 2001. Biogenic amines in wine: the role of lactic acid bacteria. FEMS Microbiology Letters. 199:9-13.
6. http://www.agsci.ubc.ca/wine/research.htm
7. Volschenk, H., Viljoen-Bloom, M., Subden, R.E., and H.J. van Vuuren. 2001. Malo-ethanolic fermentation in grape must by recombinant strains of Saccharomyces cerevisiae. Yeast. 18:963-970.
8. Sandeman, G.G. Sons & Co. 1955. Port and sherry: the story of two fine wines. George G. Sandeman Sons, London.
(Art by Jane Wang – note that high res versions of image files available here)